 Tel/WhatsApp
Tel/WhatsApp



Pyrolysis is the chemical decomposition of a condensed substance by heating. is a special case of thermolysis, and is most commonly used for organic materials. It occurs spontaneously at high temperatures (ie above 300 °C for wood, it varies for other material), for example in wildfires or when vegetation comes into contact with lava in volcanic eruptions. It does not involve reactions with oxygen or any other reagents but can take place in their presence. Extreme pyrolysis, which leaves only carbon as the residue, is called carbonization and is also related to the chemical process of charring.
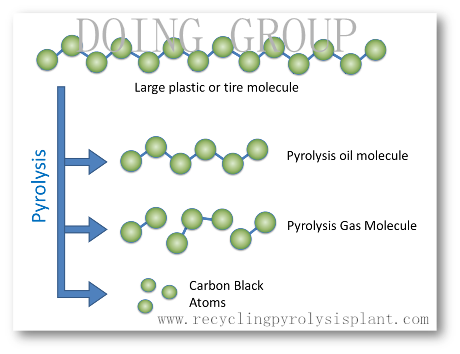
Pyrolysis
Pyrolysis is heavily used in the chemical industry, for example, to produce charcoal, activated carbon, methanol and other chemicals from wood, to convert ethylene dichloride into vinyl chloride to make PVC, to produce coke from coal, to convert biomass into syngas, to turn waste into safely disposable substances, and for the cracking of medium-weight hydrocarbons from oil to produce lighter ones like gasoline.